Abstract
Introduction: Extramedullary presentation of acute myeloid leukaemia (EM-AML) is rare, occurring in 2.5 -9.1% of patients. There is limited evidence on how to manage these patients specifically, with its impact on outcomes for patients undergoing allogeneic haematopoietic stem cell transplant (AlloSCT) not well characterised (Bakst et al. Blood 2011).
Methods: A single centre retrospective analysis was conducted for patients diagnosed with EM-AML pre- or post-AlloSCT between August 2000 and June 2021 to characterise outcomes for this patient group.
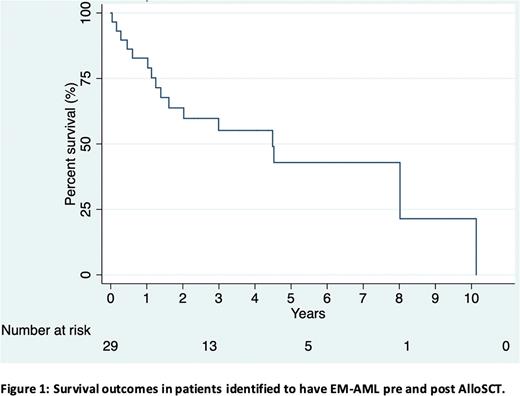
Results: 29 patients with AML (male = 18, female =11) were identified from the institutional alloSCT database. Median age at AlloSCT was 43 years (range 17-68). 76% (n=22) presented with EM-AML pre- and 24% (n=7) post-AlloSCT. 62% (n=18) had EM-AML and bone marrow involvement against 38% (n=11) with isolated EM-AML. The most common site of involvement was skin (52%; n=14). 7 patients had a FLT3 mutation and 3 had a complex karyotype at diagnosis. 55% (n=16) of patients underwent AlloSCT in CR1, 21% (n=6) in CR2, 3% (n=1) in CR3, 14% (n=4) in relapse. Remission status for one patient was not recorded and one was untreated pre-AlloSCT. 66% (n=19) underwent myeloablative conditioning and 41% (n=10) reduced intensity conditioning. Donor type included matched sibling donor (48%; n=14), matched Unrelated Donor (34%; n=10), Haplo-identical donor (14%; n=4) and umbilical cord (2%; n=1). 52% (n=15) experienced acute graft-versus-host disease (GvHD) and 62% chronic GvHD (n=18). Median overall survival (OS) post AlloSCT was 24 months (Range 1-120). Median OS for patients in CR1 pre-AlloSCT was 28 months vs 53 months for patients in CR2. The most common cause of death was progressive disease (36%). Median Relapse Free survival (RFS) post AlloSCT was 15 months (Range 1-116). Of the 7 patients who relapsed with EM-AML post AlloSCT only 1 remains alive at 49 months.
Conclusion: Our retrospective series confirms the poor outcomes associated with EM-AML despite AlloSCT with a median OS of 24 months. Additionally, patients who relapse post-allograft with EM-AML have an abysmal outcome with the majority dying of progressive disease. New treatment strategies are therefore needed to improve outcomes in this difficult patient group.
Disclosures
Bajel:Abbvie: Honoraria; Amgen: Honoraria, Speakers Bureau; Astellas: Honoraria; Novartis: Honoraria; Pfizer: Honoraria; Takeda: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal